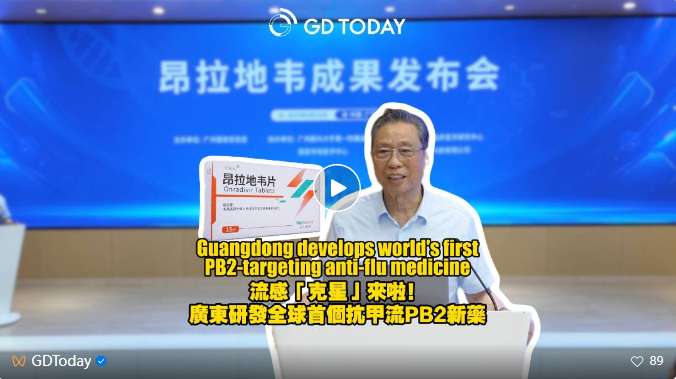
Guangdong develops world's first PB2-targeting anti-flu medicine
The Guangzhou National Laboratory has spearheaded the development of Onradivir tablets,the world's first innovative drug targeting the PB2subunit of the influenza A virus RNA polymerase,which demonstrates a40%faster reduction in symptoms in clinical trials compared to placebo.
The drug has been approved for marketing by China's National Medical Products Administration in May.
Significant efficacy
Relevant researchers indicate that Onradivir boasts remarkable advantages of rapid action,strong potency,and low resistance,promising to elevate the overall level of influenza prevention and treatment in China and globally.
Research data reveal that after taking Onradivir,patients experience a rapid decline in viral load.In vitro antiviral activity studies show a nearly40%reduction in symptom relief time compared to the placebo group(untreated group).In terms of fever reduction,the time is shortened by nearly39%compared to the placebo group.
Notably,Onradivir exhibits a low rate of resistant mutations,with no suspected resistance cases in Phase II clinical trials and a1.6%incidence in Phase III.It is also effective against"super flu viruses"resistant to some other marketed anti-influenza drugs.
Furthermore,Onradivir offers"multi-purpose"benefits,being effective against highly pathogenic avian influenza viruses such as H7N9and H5N6,thus providing a tool for responding to avian-to-human influenza outbreaks.
Providing a Chinese solution for global influenza control
Globally,there are approximately1billion influenza cases annually,including3to5million severe cases and290,000to650,000deaths.The influenza season during winter and spring imposes a significant public health burden worldwide.
Onradivir,as a new drug targeting the PB2subunit,employs a"cap-snatching"mechanism(seizing the mRNA cap of host cells),distinct from Baloxavir's"endonuclease cleavage"mechanism(cutting host mRNA).Previous attempts by foreign institutions to develop drugs using this mechanism did not achieve final success in clinical studies.
As the world's first innovative drug targeting the PB2subunit,Onradivir tablets offer a novel target for drug-resistant viruses,representing a Class1innovative drug of international standard from China.
The drug has been granted patent authorizations in multiple countries,including China and the United States.Preclinical basic pharmacological studies,as well as results from Phase II and III clinical trials,have been published in journals such as Drugs,The Lancet Infectious Diseases,and The Lancet Respiratory Medicine.
Zhong Nanshan,Academician of the Chinese Academy of Engineering and Director of the Guangzhou National Laboratory,stated that Onradivir's contribution lies not only in breakthroughs in drug efficacy and safety but also in providing a Chinese solution for global influenza control.
"Now we are very confident that it can be better than other international drugs and even more affordable,"said Zhong.
 最新热点
最新热点
-
美伊“接近结束战事”传言刺激国际油价波动
-
交通部门细化服务 提升假期出行质量
交通部门细化服务 提升假期出行质量
最新热点央视网消息(新闻联播):记者从交通运输部了解到,5月3日,预计全社会跨区域人员流动量超2.9亿人次,同比增长2.5%。探亲、旅游、休闲等出行需求继续保持旺盛...
-
德国外长:伊朗应立即开放霍尔木兹海峡
德国外长:伊朗应立即开放霍尔木兹海峡
最新热点德国外交部长瓦德富尔3日表示,伊朗应立即开放霍尔木兹海峡。 瓦德富尔当天在社交媒体上发文说,德国和美国在相关问题上拥有共同目标,即美国国务卿鲁比奥所要...
-
时政专题片丨为有源头活水来——习近平总书记指引基础研究高质量发展
时政专题片丨为有源头活水来——习近平总书记指引基础研究高质量发展
最新热点4月30日上午,习近平总书记在上海出席加强基础研究座谈会并发表重要讲话,为新形势下加强基础研究指明前进方向、提供根本遵循。 党的十八大以来,以习近...
-
欧佩克国际发展基金启动15亿美元援助计划
欧佩克国际发展基金启动15亿美元援助计划
最新热点欧佩克国际发展基金29日宣布,已启动一项15亿美元援助计划,旨在帮助伙伴国家应对中东战事引发的能源、大宗商品及贸易中断带来的经济挑战。 欧佩克国际发展...

















 法国华侨华人会第二十五届主席团就职典礼成功举办
法国华侨华人会第二十五届主席团就职典礼成功举办
 八方游客聚昆明石林赴“阿诗玛之约”
八方游客聚昆明石林赴“阿诗玛之约”
 借亚沙会东风 三亚多举措吸引国际游客
借亚沙会东风 三亚多举措吸引国际游客
 第六届消博会收官 参展专业采购商人数创新高
第六届消博会收官 参展专业采购商人数创新高
